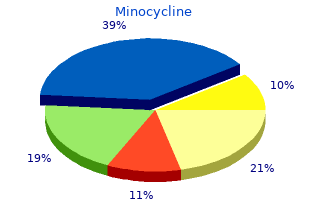
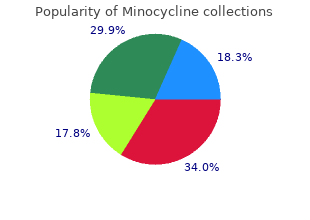
Minocycline
"Generic 50 mg minocycline overnight delivery, antibiotics for acne and depression."
By: Paul Reynolds, PharmD, BCPS
- Critical Care Pharmacy Specialist, University of Colorado Hospital
- Clinical Assistant Professor, Department of Clinical Pharmacy, Skaggs School of Pharmacy and Pharmaceutical Sciences, University of Colorado, Aurora, Colorado

http://www.ucdenver.edu/academics/colleges/pharmacy/Departments/ClinicalPharmacy/DOCPFaculty/Q-Z/Pages/Paul-Reynolds,-PharmD.aspx
Often antimicrobial bath rug purchase genuine minocycline line, overexpression of a gene that regulates the biosynthetic cluster of an antibiotic increases titers antibiotics quiz nursing buy minocycline 50 mg low price. For instance strong antibiotics for sinus infection order online minocycline, overexpression of sanG bacterial endospore order minocycline australia, a transcriptional regulator of the nikkomycin biosynthetic genes in Streptomyces ansochromogenes, increased titers of the antibiotic (3). In actinomycetes, synthesis of antibiotics often coincides with complex morphological changes during the life cycle. Compounds produced during these stages of differentiation comprise nearly two-thirds of bioactive molecules synthesized by microorganisms, including antibiotics, antitumor agents, and immunosuppressants. Apart from the pathway-specific regulators for antibiotic biosynthesis, other genes of S. For example, mutation or deletion of bldD causes defects both in the formation of aerial hyphae and the production of antibiotics (7). To our knowledge, none of the genes with characterized roles in the development of S. The altered and coordinated expression of nearly all of the ery genes suggested the existence of a common regulator for the ery cluster, although the genome sequence of S. In this study, we purified and identified a regulatory protein, BldD, which binds to all of the promoters in the ery cluster. This work reveals multiple cellular roles of BldD and makes progress toward the rational manipulation of actinomycetes to overproduce antibiotics. Results and Discussion Protein Binding to the Promoters of the Erythromycin Biosynthetic Gene Cluster. Several tests indicated that the protein(s) causing the shifts bound to ery promoters specifically. The predicted BldD sequence yielded strong matches to the BldD homologs of several actinomycetes. Analysis of sequences for Aeromicrobium erythreum, which also synthesizes erythromycin, and Micromonospora megalomicea, which synthesizes the related molecule megalomicin, suggests that the ery cluster once might have contained a regulatory gene. Both species have biosynthetic gene clusters extremely similar to the ery cluster in S. The numbers above the genes are gene numbers in the genome sequence for both species. Combined with the bald phenotype of the bldD mutant (see above), this synteny reinforces the idea that S. When administered concurrently with penicillin, which medication diminishes the bactericidal effect? Focus on what matters Your fellow students write the study notes themselves, which is why the documents are always reliable and up-to-date. Hawker F "Five cases of pulmonary oedema associated with beta 2-sympathomimetic treatment of premature labour. Am J Med 113 (2002): 701-2Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances. Ships to Russian Federation Save 5% more with Subscribe & Save Ships to Russian Federation Ships to Russian Federation Save more with Subscribe & Save Ships to Russian Federation Save more with Subscribe & Save Ships to Russian Federation Save 10% more with Subscribe & Save Ships to Russian Federation Save 10% more with Subscribe & Save Ships to Russian Federation Best Seller in Antifungal Remedies 16,095 Only 1 left in stock - order soon. Save more with Subscribe & Save 5% coupon applied at checkout Save 5% with coupon Ships to Russian Federation Save 5% more with Subscribe & Save Ships to Russian Federation Ships to Russian Federation Ships to Russian Federation Ships to Russian Federation Save 10% more with Subscribe & Save $0. Gennimatas”, Athens, Greece †Section of Cardiovascular Medicine, Yale University School of Medicine, Yale University, New Haven, Connecticut ‡1st Department of Cardiology, Athens University School of Medicine, Athens, Greece ↵∗Reprint requests and correspondence: Dr. It is commonly classified as an anti-inflammatory agent, although its mechanism of action does not involve the arachidonic acid pathway affected by non-steroid anti-inflammatory drugs and glucocorticoids. Colchicine inhibits microtubule polymerization by binding to tubulin, thus affecting any process that requires cytoskeletal changes, including cell mitosis and neutrophil motility. Recent studies suggest that colchicine may prove to be useful in a much wider spectrum of cardiovascular diseases than previously suspected, rekindling the interest in this old drug. In this review we briefly present the biochemical characteristics, mechanism of action and side-effects of colchicine, as well as examine what is currently known about the promising role of colchicine in cardiovascular medicine beyond pericardial disease. Key WordsColchicine, the extract from the Colchicum autumnale plant (autumn crocus), used by ancient Greeks more than 2,000 years ago (1), is one of the oldest known drugs still prescribed today. Caventou were the first to extract colchicine as the active alkaloid of colchicum (2). Colchicine is widely used in in vitro experiments requiring cytoskeleton disruption. In this review we refer to studies claiming a wider role for colchicine in cardiovascular disease, beyond traditional indications in pericarditic syndromes, as well as examine whether our current knowledge about colchicine mechanisms of action can offer insights relevant to clinical findings. It is true that data from really large, randomized trials of colchicine for potential novel indications in cardiovascular disease with hard clinical endpoints are lacking. As a result, a true systematic review or a meta-analysis (with the statistical processing that these terms imply) is implausible at the moment. It is clear from Table 1 that a number of well-planned and well-conducted studies have been published. For these reasons, the scope of the present review encompasses 2 main goals: 1) presenting an exhaustive account of clinical studies indicating potential new uses of colchicine in heart disease; and 2) stimulating interest among cardiovascular specialists for this old and, possibly, underutilized medicine. Table 1 Important Clinical Studies Examining the Role of Colchicine Administration in Cardiovascular Disease, Beyond Pericarditic Syndromes Pharmacokinetics Colchicine is a lipid-soluble drug with a [pKa] = 12. After oral ingestion it has a 44% bioavailability and binds relatively weakly to albumin (32% bound) (6). Its peak plasma concentration occurs in about 1 h, with a long terminal half-life (5). Colchicine enters red blood cells and leucocytes where it remains in much higher concentrations than the plasma (6). Colchicine can be found inside leukocytes for up to several days after administration (7), with this local pooling and long half-life explaining why a once- or twice-daily dosage scheme is enough for inflammation suppression. Peak plasma concentration and plasma half-life can be significantly prolonged in patients with liver or renal failure or in the elderly (9), with side effects appearing even at low doses. Colchicine has a relatively low therapeutic index, with effective steady-state plasma concentrations ranging between 0. A significant percentage of patients report adverse effects, mainly gastrointestinal symptoms, serious enough to cause discontinuation of therapy even at low daily doses. Rare side effects of chronic colchicine administration include liver failure, bone marrow depression, which may manifest as leucopenia or aplastic anemia, and rhabdomyolysis (10). Mechanisms of Action Due to the rapid suppression of gouty arthritis without interfering with uric acid kinetics, colchicine is generally classified as an anti-inflammatory agent in the medical literature. Colchicine binds to unpolymerized tubulin heterodimers, forming a stable complex that effectively inhibits microtubule dynamics upon binding to microtubule ends. It causes microtubule depolymerization by inhibiting lateral contacts between protofilaments (11), thus affecting any process that requires cytoskeletal changes, including cell mitosis, exocytosis, and neutrophil motility (8). Microtubule structure is illustrated in Figure 1, while the spatial interaction of colchicine and tubulin subunits is illustrated in Figure 2. At low concentrations, colchicine inhibits the formation of microtubules while at higher concentrations it promotes their depolymerization (Fig. Colchicine has been found to impair adhesion of neutrophils to the vascular endothelium. In vitro experiments have shown that colchicine diminishes endothelial selectin family-dependent adhesiveness, affecting both endothelial E-selectin and, at higher concentrations, neutrophil L-selectin surface expression (12). Thirteen protofilaments are linked laterally, forming the 3-dimensional cylinder of the microtubule with β-tubulin exposed at the microtubule plus (+) end (yellow circles) and α-tubulin exposed at the microtubule minus (–) end (red circles). Docking and hydropathic scoring of polysubstituted pyrrole compounds with antitubulin activity. Theoretically, this combination may be inexpedient, as the bacteriostatic agent may antagonize the effect of the bactericidal agent. In vitro time–kill curves were generated with clinically relevant concentrations of penicillin (10 mg/L) and erythromycin (1 mg/L), either individually or in combination. Antagonism between penicillin and erythromycin was observed for the four isolates. After intraperitoneal inoculation, penicillin and erythromycin were given either individually or in combination. For two of the four isolates, mortality was significantly higher in the groups treated with the combination of penicillin and erythromycin than in the groups treated with penicillin alone [32/36 (86%) vs 3/12 (25%), P P in vivo time–kill curves showed that there was antagonism between erythromycin and penicillin for the examined isolate. The antagonism demonstrated in vitro and in vivo between penicillin and erythromycin suggests that β-lactam antibiotics and macrolides should not be administered together unless pneumococcal infection is ruled out. Introduction It is a challenge to choose the right therapy for patients with pneumonia, as the aetiology cannot be predicted based on the symptoms and clinical findings alone, and a final microbiological diagnosis has usually not been established before treatment has to start. Streptococcus pneumoniae is the most frequently isolated organism in community-acquired pneumonia, especially in small children and older patients, and the infection still carries a high mortality.
Chloride channels are often increased on the surface of malignant cells virus spreading minocycline 50mg with visa, compared with normal cells bacteria encyclopedia generic 50mg minocycline free shipping, potentially making them more sensitive to virus 7th grade science purchase minocycline 50 mg mastercard alterations in chloride flux by ivermectin virus in colorado cheap minocycline 50 mg with amex. Alterations in intracellular chloride concentrations also affect basic homeostatic parameters, such as intracellular Ca2+ levels, pH, and cell volume,38 and alteration of these parameters can induce apoptosis. Consequently, we evaluated the combination of these drugs with ivermectin and demonstrated synergy with both of these drugs. Given its prior safety record in humans and animals coupled with its preclinical efficacy in leukemia, a phase 1 clinical trial could be conducted to determine the tolerance and biologic activity of oral ivermectin in these patients. The publication costs of this article were defrayed in part by page charge payment. Conflict-of-interest disclosure: The authors declare no competing financial interests. Clioquinol inhibits the proteasome and displays preclinical activity in leukemia and myeloma. Comparative evaluation of acute toxicity of ivermectin by two methods after single subcutaneous administration in rats. Safety, tolerability, and pharmacokinetics of escalating high doses of ivermectin in healthy adult subjects. Disruption of the endoplasmic reticulum and increases in cytoplasmic calcium are early events in cell death induced by the natural triterpenoid, Asiatic acid. The dietary isothiocyanate, sulforaphane, targets pathways of apoptosis, cell cycle arrest, and oxidative stress in human pancreatic cancer cells and inhibits tumor growth in severe combined immunodeficient mice. Chelation of intracellular iron with the antifungal agent, ciclopirox olamine, induces cell death in leukemia and myeloma cells. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. The hyperpolarizing impact of glycine on endothelial cells may be anti-atherogenic. Digoxin- and monensin-induced changes of intracellular Ca2+ concentration in isolated guinea-pig ventricular myocyte. Influence of frequency of stimulation on the toxicity of digoxin on isolated guinea-pig atria in different extracellular Ca2+. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Involvement of oxygen radicals in cytarabine-induced apoptosis in human polymorphonuclear cells. Mansonella perstans: safety and efficacy of ivermectin alone, albendazole alone, and the two drugs in combination. Monitoring the efficacy of ivermectin and albendazole against gastro intestinal nematodes of cattle in Northern Europe. Ivermectin distribution in the plasma and tissues of patients infected with Onchocerca volvulus. Crusted scabies in an adult T-cell leukemia/lymphoma patient successfully treated with oral ivermectin. Comparison of the sensitivity of normal and leukaemic myeloid progenitors to in-vitro incubation with cytotoxic drugs: a study of pharmacological purging. Chemotherapeutic sensitivity of normal and leukemic hematopoietic progenitor cells to N-[4-(9-acridinylamino)-3-methoxyphenyl]-methanesulfonamide, a new anticancer agent. Mercuric chloride-induced reactive oxygen species and its effect on antioxidant enzymes in different regions of rat brain. Expression of myeloperoxidase enhances the chemosensitivity of leukemia cells through the generation of reactive oxygen species and the nitration of protein. Amoxicillin and clavulanate combination is an antibiotic that belongs to the group of medicines known as penicillins and beta-lactamase inhibitors. This product is available in the following dosage forms: Tablet, Chewable Tablet Powder for Suspension Tablet, Extended Release Before Using In deciding to use a medicine, the risks of taking the medicine must be weighed against the good it will do. Pediatric Appropriate studies performed to date have not demonstrated pediatric-specific problems that would limit the usefulness of amoxicillin and clavulanate chewable tablets and oral suspension in children. Appropriate studies performed to date have not demonstrated pediatric-specific problems that would limit the usefulness of amoxicillin and clavulanate tablets in children weighing 40 kilograms (kg) or more. However, safety and efficacy have not been established in children weighing less than 40 kg. Appropriate studies have not been performed on the relationship of age to the effects of amoxicillin and clavulanate extended-release tablets in teenagers and children younger than 16 years of age. Geriatric Appropriate studies performed to date have not demonstrated geriatric-specific problems that would limit the usefulness of amoxicillin and clavulanate combination in the elderly. However, elderly patients are more likely to have age-related kidney problems, which may require an adjustment in the dose for patients receiving amoxicillin and clavulanate combination. Make sure you tell your doctor if you have any other medical problems, especially: Diarrhea or Liver disease—Use with caution. Kidney disease, severe—The extended-release tablets should not be used in patients with this condition. Liver disease (caused by Augmentin®), history of or Mononucleosis ("mono")—Should not be used in patients with these conditions. However, it is best to take this medicine at the start of a meal or snack to avoid an upset stomach. If you are using the chewable tablets, chew the tablet completely before swallowing. There are certain flavors that can be mixed with the oral suspension to make it taste better. If you or your child do not like the taste of this medicine, ask your pharmacist about these flavorings. For bacterial infections: For oral dosage forms (chewable tablets or suspension): Adults, teenagers, and children weighing 40 kilograms (kg) or more—250 to 500 milligrams (mg) every 8 hours, or 500 to 875 mg every 12 hours. Children 3 months of age and older and weighing less than 40 kilograms (kg)—Dose is based on body weight and must be determined by your doctor. The dose is usually 20 to 40 mg per kg of body weight per day, divided and given every 8 hours, or 25 to 45 mg per kg of body weight per day divided and given every 12 hours. Children younger than 3 months of age—Dose is based on body weight and must be determined by your doctor. The dose is usually 30 mg per kg of body weight per day, divided and given every 12 hours. For oral dosage form (extended-release tablets): Adults—2000 milligrams (mg) every 12 hours. For oral dosage form (tablets): Adults, teenagers, and children weighing 40 kilograms (kg) or more—250 to 500 milligrams (mg) every 8 hours, or 500 to 875 mg every 12 hours. Children weighing less than 40 kg—Use and dose must be determined by your doctor. Store the chewable tablets, regular tablets, and extended-release tablets at room temperature, away from heat, moisture, and direct light. Your doctor will check the progress of you or your child while you are using this medicine. Call your doctor right away if you or your child have itching, hives, hoarseness, trouble breathing, trouble swallowing, or any swelling of your hands, face, or mouth after taking this medicine. Amoxicillin and clavulanate combination may cause diarrhea, and in some cases it can be severe. If you have any questions or if mild diarrhea continues or gets worse, check with your doctor. Amoxicillin and clavulanate combination may decrease the effects of some oral contraceptives (birth control pills). Before you or your child have any medical tests, tell the doctor in charge that you are taking this medicine. Check with your doctor immediately if any of the following side effects occur: Less common Hives or welts itching itching of the vagina or genital area pain during sexual intercourse redness of the skin skin rash thick, white vaginal discharge with no odor or with a mild odor Rare Bloody or cloudy urine fever greatly decreased frequency of urination or amount of urine seizures swelling of the feet or lower legs Incidence not known Abdominal or stomach cramps or tenderness back, leg, or stomach pains black, hairy tongue black, tarry stools bleeding gums blistering, peeling, or loosening of the skin bloating blood in the stools bloody nose chest pain chills clay-colored stools cough or hoarseness cracks in the skin dark urine diarrhea diarrhea, watery and severe, which may also be bloody difficulty with breathing difficulty with moving difficulty with swallowing dizziness fast heartbeat fever with or without chills general body swelling general feeling of tiredness or weakness headache heavier menstrual periods increased thirst joint or muscle pain large, hive-like swelling on the face, eyelids, lips, tongue, throat, hands, legs, feet, or sex organs light-colored stools loss of appetite loss of heat from the body lower back or side pain muscle stiffness nausea or vomiting pain pain, swelling, or redness in the joints painful or difficult urination pale skin pinpoint red spots on the skin puffiness or swelling of the eyelids or around the eyes, face, lips, or tongue rash red skin lesions, often with a purple center red, irritated eyes red, swollen skin scaly skin sore throat sores, ulcers, or white spots on the lips or in the mouth swollen glands tightness in the chest troubled breathing with exertion unpleasant breath odor unusual bleeding or bruising unusual tiredness or weakness unusual weight loss upper right abdominal or stomach pain vomiting of blood white patches in the mouth or throat or on the tongue white patches with diaper rash yellow eyes or skin Get emergency help immediately if any of the following symptoms of overdose occur: Symptoms of overdose Abdominal or stomach pain cloudy urine diarrhea greatly decreased frequency of urination or amount of urine sleepiness Some side effects may occur that usually do not need medical attention. Check with your health care professional if any of the following side effects continue or are bothersome or if you have any questions about them: Rare Anxiety dry mouth hyperventilation irregular heartbeats irritability restlessness shaking sleeplessness tooth discoloration trouble sitting still trouble with sleeping Incidence not known Burning feeling in the chest or stomach indigestion redness, swelling, or soreness of the tongue stomach upset swelling or inflammation of the mouth Other side effects not listed may also occur in some patients. Adults and children who weigh more than 40 kg (88 lb): 250 mg (based on amoxicillin component) P.
Discount minocycline 50mg online. Antimicrobial medication - Medical Definition.
Melaleuca viridiflora (Niauli Oil). Minocycline.
- Cough and inflammation of the airways.
- What is Niauli Oil?
- Are there safety concerns?
- Dosing considerations for Niauli Oil.
- How does Niauli Oil work?
Source: http://www.rxlist.com/script/main/art.asp?articlekey=96576
After thorough mixing medicine for lower uti discount minocycline 50 mg without a prescription, the samples were centrifuged at 11 virus clothing generic minocycline 50mg free shipping,000 rpm for 5 min at approximately +4°C antibiotic resistance week order minocycline once a day. Following centrifugation and dilution of the supernatant with 5 mM ammonium acetate buffer antibiotics review pdf generic 50mg minocycline visa, 20 μl of each sample was injected. Human microdialysate samples (10 μl) were diluted by 90 μl of ammonium acetate buffer (5 mM) containing the corresponding internal standard. Samples with concentration above the upper quantification limit were prediluted with isotonic saline solution. Under these conditions, erythromycin, anhydroerythromycin, and the internal standard were eluted after approximately 1. Plasma samples were measured against a calibration curve prepared in human drug-free plasma. Calibration standards were prepared by adding the defined amounts of standard solution of analyte to drug-free plasma. Calibration standards were prepared by adding the defined amounts of standard solution of analyte to 0. The intraday precision (accuracy) for erythromycin A and anhydroerythromycin A in human plasma was determined as 0. The interday precision (accuracy) for erythromycin A and anhydroerythromycin A in human plasma was determined as 2. Cavg was defined as the average concentration observed at the three sampling points on day 5. The free fraction of erythromycin in plasma was estimated to be 10% based on data derived from the literature (41). For comparison of parameters between compartments, Wilcoxon matched-pairs test was employed. Erythromycin was well tolerated, and all six subjects completed the study according to the protocol. Gastric pain, pain at microdialysis probe insertion, loss of appetite, meteorism, leucocytopenia, and thrombocytopenia were observed in one subject each; all adverse events were considered mild, and no serious adverse events were reported. The comparative mean concentration-time profiles of erythromycin during one dosing interval of 6 h are shown in Fig. For all compartments, higher mean concentrations of erythromycin were observed on day 3 of active treatment in comparison to day 1. Two days after the end of treatment, a significant decrease of mean erythromycin concentrations was present, while after 7 days the erythromycin concentration was under the limit of detection in all compartments. While total concentrations in plasma exceeded concentrations in interstitial space of soft tissues by approximately 10-fold, the free fraction of erythromycin in plasma was in good agreement with concentrations in muscle and subcutaneous adipose tissue. In vitro cellular uptake of erythromycin was previously extensively investigated (5, 24, 42). Active and passive transport through cell membranes resulting in accumulation of erythromycin in the intracellular compartment has been described previously (42). The cellular/extracellular ratios were constant over a wide range of concentrations (24). Independent of the cell type, antimicrobial active erythromycin rapidly effused when cells were incubated in antibiotic-free medium. Thus, the observed accumulation of erythromycin of approximately 40-fold compared to plasma is in good agreement with previous data (22, 26). However, a limitation of the present study is that all investigations were performed in healthy, noninflammed tissues. On the other hand, acidification leads to a striking reduction of antimicrobial activity of erythromycin, so the overall impact of major local inflammation remains unclear (17). For many inflammatory airway diseases like diffuse panbronchiolitis or cystic fibrosis, the immune-modulatory effect of erythromycin is considered responsible for beneficial effects rather than its antimicrobial action (12). It has to be noted that the observed thresholds were obtained by calculations from plasma and therefore may not be readily transferred to other compartments. Compared to newer macrolides, the relatively short period of subinhibitory concentrations may be advantageous (23). Higher initial doses of erythromycin might be considered to accelerate sufficient free concentrations in tissue. Evolving resistance patterns of Streptococcus pneumoniae: a link with long-acting macrolide consumption? Macrolide and azithromycin use are linked to increased macrolide resistance in Streptococcus pneumoniae. Cellular uptake and efflux of azithromycin, erythromycin, clarithromycin, telithromycin, and cethromycin. Clinical relevance of intracellular and extracellular concentrations of macrolides. Kinetics of erythromycin uptake and release by human lymphocytes and polymorphonuclear leucocytes. Pharmacoepidemiological analysis of provincial differences between consumption of macrolides and rates of erythromycin resistance among Streptococcus pyogenes isolates in Spain. Comparison of outpatient systemic antibacterial use in 2004 in the United States and 27 European countries. Outpatient antibiotic use in Europe and association with resistance: a cross-national database study. Effect of carbon dioxide and pH on susceptibility of Bacteroides fragilis group to erythromycin. In vitro antibacterial activities of S-013420, a novel bicyclolide, against respiratory tract pathogens. Antibacterial activities of erythromycins A, B, C, and D and some of their derivatives. Comparison of microbiologic and high-performance liquid chromatography assays to determine plasma concentrations, pharmacokinetics, and bioavailability of erythromycin base in plasma of foals after intravenous or intragastric administration. Effect of azithromycin and clarithromycin therapy on pharyngeal carriage of macrolide-resistant streptococci in healthy volunteers: a randomised, double-blind, placebo-controlled study. Uptake, accumulation, and egress of erythromycin by tissue culture cells of human origin. Antimicrobial drug prescription in ambulatory care settings, United States, 1992–2000. Erythromycin uptake and accumulation by human polymorphonuclear leukocytes and efficacy of erythromycin in killing ingested Legionella pneumophila. Penetration of ciprofloxacin into the interstitial space of inflamed foot lesions in non-insulin-dependent diabetes mellitus patients. Mean cell volume of human blood leucocytes and resident and activated murine macrophages. Pharmacokinetics of antibiotics in natural and experimental superficial compartments in animals and humans. Efficacy and tolerability of erythromycin acistrate and erythromycin stearate in acute skin infections of patients with atopic eczema. Influence of pharmacokinetics/pharmacodynamics of antibacterials in their dosing regimen selection. The effect of changes in the consumption of macrolide antibiotics on erythromycin resistance in group A streptococci in Finland. The inhibition of drug oxidation by anhydroerythromycin, an acid degradation product of erythromycin. Pharmacokinetics of single- and multiple-dose oral clarithromycin in soft tissues determined by microdialysis. Comparative efficacy of 5 days of dirithromycin and 7 days of erythromycin in skin and soft tissue infections. Lung microdialysis-a powerful tool for the determination of exogenous and endogenous compounds in the lower respiratory tract (mini-review). Relevance of soft-tissue penetration by levofloxacin for target site bacterial killing in patients with sepsis. We always aim to dispatch all Royal Mail orders received before 15:00 the same working day. We ensure all parcels are in plain packaging for discreet delivery of your medicines and pharmacy goods. You should notify our customer support team if it is not safe for the package to be posted through the letterbox, for example due to animals or small children.
Guanfacine: (Major) Ciprofloxacin may significantly increase guanfacine plasma concentrations antibiotic resistance who 2011 order minocycline 50mg otc. Halothane: (Major) Halogenated anesthetics should be used cautiously and with close monitoring with ciprofloxacin antibiotic resistance marker purchase 50mg minocycline. Homatropine; Hydrocodone: (Moderate) Consider a reduced dose of hydrocodone with frequent monitoring for respiratory depression and sedation if concurrent use of ciprofloxacin is necessary bacteria definition order 50mg minocycline fast delivery. Hydrocodone: (Moderate) Consider a reduced dose of hydrocodone with frequent monitoring for respiratory depression and sedation if concurrent use of ciprofloxacin is necessary antibiotics walking pneumonia cheap 50mg minocycline otc. Hydrocodone; Ibuprofen: (Moderate) Consider a reduced dose of hydrocodone with frequent monitoring for respiratory depression and sedation if concurrent use of ciprofloxacin is necessary. Hydrocodone; Phenylephrine: (Moderate) Consider a reduced dose of hydrocodone with frequent monitoring for respiratory depression and sedation if concurrent use of ciprofloxacin is necessary. Hydrocodone; Potassium Guaiacolsulfonate; Pseudoephedrine: (Moderate) Consider a reduced dose of hydrocodone with frequent monitoring for respiratory depression and sedation if concurrent use of ciprofloxacin is necessary. Close monitor patients for any signs of toxicity, such as meylosuppression, fluid retention, and bleeding. Indinavir: (Moderate) Use caution with the coadministration of ciprofloxacin and indinavir. The plasma concentrations of indinavir may be elevated when administered concurrently with ciprofloxacin. Insulin Degludec: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including insulin, are coadministered. Insulin Glargine: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including insulin, are coadministered. Insulin Glulisine: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including insulin, are coadministered. Insulin Lispro; Insulin Lispro Protamine: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including insulin, are coadministered. Insulins: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including insulin, are coadministered. Isavuconazonium: (Moderate) Concomitant use of isavuconazonium with ciprofloxacin may result in increased serum concentrations of isavuconazonium. Isoflurane: (Major) Halogenated anesthetics should be used cautiously and with close monitoring with ciprofloxacin. Ivabradine: (Major) Avoid coadministration of ivabradine and ciprofloxacin as increased concentrations of ivabradine are possible. Lemborexant: (Major) Avoid coadministration of lemborexant and ciprofloxacin as concurrent use is expected to significantly increase lemborexant exposure and the risk of adverse effects. Lidocaine; Prilocaine: (Moderate) Concomitant use of systemic lidocaine and ciprofloxacin may increase lidocaine plasma concentrations by decreasing lidocaine clearance and therefore prolonging the elimination half-life. Linagliptin; Metformin: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including dipeptidyl peptidase-4 inhibitors, are coadministered. Lithium: (Moderate) Lithium should be used cautiously and with close monitoring with ciprofloxacin. Lumateperone: (Major) Avoid coadministration of lumateperone and ciprofloxacin as concurrent use may increase lumateperone exposure and the risk of adverse effects. Magnesium Citrate: (Major) Administer oral ciprofloxacin at least 2 hours before or 6 hours after magnesium citrate. Magnesium Salicylate: (Major) Administer oral ciprofloxacin at least 2 hours before or 6 hours after magnesium salicylate. Magnesium Salts: (Major) Administer oral ciprofloxacin at least 2 hours before or 6 hours after oral products that contain magnesium. Meglitinides: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including meglitinides, are coadministered. This may potentially lead to increased methotrexate plasma concentrations and increase the risk of methotrexate associated toxic reactions. Metoclopramide: (Minor) Metoclopramide accelerates the absorption of oral ciprofloxacin. This results in shorter time to reach maximum ciprofloxacin plasma concentrations. In 6 cancer patients receiving chemotherapy and after 13 days of chemotherapy, there were decreases in mean maximum serum concentration, in mean time to reach maximum concentration, and in the area under the concentration curve of ciprofloxacin. In addition to a rise in predose concentrations with continued antibiotic use, some patients did not have a large reduction in their predose concentration. Naloxegol: (Major) Avoid concomitant administration of naloxegol and ciprofloxacin due to the potential for increased naloxegol exposure. Neratinib: (Major) Avoid concomitant use of ciprofloxacin with neratinib due to an increased risk of neratinib-related toxicity. Nisoldipine: (Major) Avoid coadministration of nisoldipine with ciprofloxacin due to increased plasma concentrations of nisoldipine. If coadministration is unavoidable, monitor blood pressure closely during concurrent use of these medications. Olaparib: (Major) Avoid coadministration of olaparib with ciprofloxacin due to the risk of increased olaparib-related adverse reactions. If concomitant use is unavoidable, reduce the dose of olaparib to 150 mg twice daily; the original dose may be resumed 3 to 5 elimination half-lives after ciprofloxacin is discontinued. Ozanimod initiation may result in a transient decrease in heart rate and atrioventricular conduction delays. Pemigatinib: (Major) Avoid coadministration of pemigatinib and ciprofloxacin due to the risk of increased pemigatinib exposure which may increase the risk of adverse reactions. If ciprofloxacin is discontinued, increase the pexidartinib dose to the original dose after 3 plasma half-lives of ciprofloxacin. Pioglitazone: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including thiazolidinediones, are coadministered. Monitor for adverse effects of pirfenidone, like elevated hepatic enzymes, arthralgia, or nausea. Polyethylene Glycol; Electrolytes: (Major) Administer quinolones at least 2 hours before or 6 hours after administration of magnesium sulfate; potassium sulfate; sodium sulfate. Polyethylene Glycol; Electrolytes; Ascorbic Acid: (Major) Administer quinolones at least 2 hours before or 6 hours after administration of magnesium sulfate; potassium sulfate; sodium sulfate. Polysaccharide-Iron Complex: (Moderate) Administer oral ciprofloxacin at least 2 hours before or 6 hours after oral products that contain iron. Examples of compounds that may interfere with quinolone bioavailability include multivitamins that contain iron. If concomitant use is unavoidable, decrease the pomalidomide dose to 2 mg once daily and monitor for pomalidomide adverse events. Prednisolone: (Moderate) Quinolones have been associated with an increased risk of tendon rupture requiring surgical repair or resulting in prolonged disability; this risk is further increased in those receiving concomitant corticosteroids. Pyridoxine, Vitamin B6: (Moderate) Administer oral ciprofloxacin at least 2 hours before or 6 hours after oral products that contain calcium. If ciprofloxacin must be administered with ramelteon, monitor the patient closely for toxicity due to elevated ramelteon serum concentrations. If ciprofloxacin is discontinued, resume the original selumetinib dose after 3 elimination half-lives of ciprofloxacin. Additionally, concomitant use of siponimod and ciprofloxacin may increase siponimod exposure. Sitagliptin: (Moderate) Monitor blood glucose carefully when systemic quinolones and antidiabetic agents, including dipeptidyl peptidase-4 inhibitors, are coadministered. Sodium Ferric Gluconate Complex; ferric pyrophosphate citrate: (Moderate) Administer oral ciprofloxacin at least 2 hours before or 6 hours after oral products that contain iron. Sonidegib: (Major) Avoid the concomitant use of sonidegib and ciprofloxacin; sonidegib exposure may be significantly increased resulting in increased risk of adverse events, particularly musculoskeletal toxicity. If concomitant use cannot be avoided, administer ciprofloxacin for less than 14 days; monitor patients closely for adverse reactions (e. If concomitant use is unavoidable, monitor electrocardiograms and correct electrolyte abnormalities. Sucralfate: (Moderate) Administer oral ciprofloxacin at least 2 hours before or 6 hours after sucralfate.


