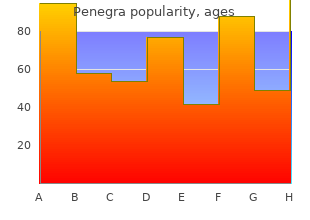
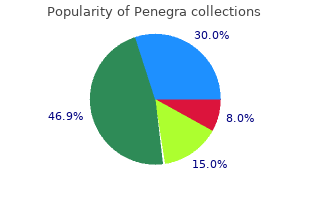
Penegra
"Purchase penegra with american express, mens health 30 minute workout."
By: Denise H. Rhoney, PharmD, FCCP, FCCM
- Ron and Nancy McFarlane Distinguished Professor and Chair, Division of Practice Advancement and Clinical Education, UNC Eshelman School of Pharmacy, Chapel Hill, North Carolina

https://pharmacy.unc.edu/news/directory/drhoney/
In addition prostate cancer nomograms purchase penegra cheap online, urine culture and susceptibility testing should be performed man healthxnet order penegra 50mg mastercard, and initial empirical therapy should be tailored and followed by (oral) administration of an appropriate antimicrobial agent on the basis of the isolated uropathogen prostate 600 plus order genuine penegra on-line. The same applies to prostate jokes buy cheap penegra 50mg ciprofloxacin and other fluoroquinolones in urological patients [158]. The choice between these agents should be based on local resistance data, and the regimen should be tailored on the basis of susceptibility results [144]. In view of the high degree of resistance, particularly among patients admitted to the department of urology, fluoroquinolones are not automatically suitable as empirical antimicrobial therapy, especially when the patient has used ciprofloxacin in the last six months [159]. Fluoroquinolones can only be recommended as empirical treatment when the patient is not seriously ill and it is considered safe to start initial oral treatment or if the patient has had an anaphylactic reaction to beta-lactam antimicrobials. If the prevalence of fluoroquinolone resistance is thought to be > 10% and the patient has 2 contraindications for third generation cephalosporins or an aminoglycoside, ciprofloxacin can be prescribed as an empirical treatment in women with uncomplicated pyelonephritis. In the event of hypersensitivity to penicillin, a third generation cephalosporin can still be prescribed, 2 with the exception of systemic anaphylaxis in the past. Only use ciprofloxacin provided that the local resistance percentages are < 10% when; Strong � the entire treatment is given orally; � patients do not require hospitalisation; � patient has an anaphylaxis for beta-lactam antimicrobials. Approximately 20% of hospital-acquired bacteraemias arise from the urinary tract, and the mortality associated with this condition is approximately 10% [160]. The incidence of bacteriuria associated with indwelling catheterisation is 3-8% per day [161-165]. Urinary catheterisation perturbs host defence mechanisms and provides easier access of uropathogens to the bladder. Indwelling urinary catheters facilitate colonisation with uropathogens by providing a surface for the attachment of host cell binding receptors recognised by bacterial adhesins, thus enhancing microbial adhesion. In addition, the uroepithelial mucosa is disrupted, exposing new binding sites for bacterial adhesins, and residual urine in the bladder is increased through pooling below the catheter bulb [168]. Recommendations Strength rating Do not carry out routine urine culture in an asymptomatic catheterised patients. The urine culture should be obtained from the freshly placed catheter prior to the initiation of antimicrobial therapy [155]. If use of the catheter can be discontinued, a culture of a voided mid-stream urine specimen should be obtained prior to the initiation of antimicrobial therapy to help guide treatment [155]. Long-term indwelling catheters should be changed at intervals adapted to the individual patient [170, 171]. Take a urine culture prior to initiating antimicrobial therapy in catheterised patients in whom Strong the catheter has been removed. Strong Treat catheter-associated asymptomatic bacteriuria prior to traumatic urinary tract Strong interventions. Strong Do not apply topical antiseptics or antimicrobials to the catheter, urethra or meatus. Results for individual trials were inconsistent with five trials including the possibility of no benefit [172]. There was no difference in infective complications recorded at up to four weeks after catheter removal. More isolates obtained from the prophylaxis group (11) were resistant to ciprofloxacin compared to the no treatment group (3) [173]. The treatment of urosepsis involves adequate life-supporting care, appropriate and prompt antimicrobial therapy, adjunctive measures and the optimal management of urinary tract disorders [176]. The decompression of any obstruction and drainage of larger infectious abscesses in the urinary tract is essential as first-line focus control [176]. Urologists are recommended to treat patients in collaboration with intensive care and infectious diseases specialists. Nosocomial urosepsis may be reduced by measures used to prevent nosocomial infection. Sepsis is diagnosed when clinical evidence of infection is accompanied by signs of systemic inflammation, presence of symptoms of organ dysfunction and persistent hypotension associated with tissue anoxia (Table 6). It is important to note that a patient can move from an almost harmless state to severe sepsis in a very short time. Mortality rates associated with severe sepsis vary depending on the organ source [177] with urinary tract sepsis generally having a lower mortality than that from other sources [178]. In recent years, the overall incidence of sepsis arising from all sources has increased by 8. Although the rate of sepsis due to fungal organisms has increased, Gram-positive bacteria have become the predominant pathogen overall. In urosepsis, as in other types of sepsis, the severity depends mostly upon the host response. Patients who are more likely to develop urosepsis include elderly patients, diabetics, immunosuppressed patients, such as transplant recipients and patients receiving cancer chemotherapy or corticosteroids. Urosepsis also depends on local factors, such as urinary tract calculi, obstruction at any level in the urinary tract, congenital uropathy, neurogenic bladder disorders, or endoscopic manoeuvres. However, all patients can be affected by bacterial species that are capable of inducing inflammation within the urinary tract. Microbiology sampling should be applied to urine, two sets of blood cultures [182], and if appropriate drainage fluids. Definition and criteria of sepsis and septic shock [14, 174, 175] Disorder Definition Sepsis Life-threatening organ dysfunction caused by a dysregulated host response to infection. Septic shock Septic shock should be defined as a subset of sepsis in which particularly profound circulatory, cellular, and metabolic abnormalities are associated with a greater risk of mortality than with sepsis alone. Patients with septic shock can be clinically identified by a vasopressor requirement to maintain a mean arterial pressure of 65 mm Hg or greater and serum lactate level greater than 2 mmol/L (>18 mg/dL) in the absence of hypovolemia. In several countries, bacterial strains can be resistant or multi-resistant and therefore difficult to treat [181]. They are molecules that regulate the amplitude and duration of the host inflammatory response. They are released from various cells including monocytes, macrophages and endothelial cells, in response to various infectious stimuli. The complex balance between proand anti-inflammatory responses is modified in severe sepsis. Mechanisms of organ failure and death in patients with sepsis remain only partially understood [178]. During severe generalised infections (bacterial, parasitic and fungal) with systemic manifestations, procalcitonin levels rise [184]. In contrast, during severe viral infections or inflammatory reactions of non-infectious origin, procalcitonin levels show only a moderate or no increase. Mid-regional proadrenomedullin has been shown to play a decisive role in the induction of hyperdynamic circulation during the early stages of sepsis and progression to septic shock [185]. Procalcitonin monitoring may be useful in patients likely to develop sepsis and to differentiate from a severe inflammatory status not due to bacterial infection [184, 186]. In addition, serum lactate is a marker of organ dysfunction and is associated with mortality in sepsis [187]. Serum lactate should therefore also be monitored in patients with severe infections. Urosepsis treatment requires a combination of treatment including treatment of the cause (obstruction of the urinary tract), adequate life-support care, and appropriate antimicrobial therapy [178, 183]. In such a situation, it is recommended that urologists collaborate with intensive care and infectious disease specialists for the best management of the patient. Antibiotic agents should be chosen according to the predominant pathogens at a given site of infection in the hospital environment. Long inpatient periods before surgery lead to a greater incidence of nosocomial infections. Antibiotic prophylaxis does not prevent stent colonisation, which appears in 100% of patients with a permanent ureteral stent and in 70% of those temporarily stented. The potential side-effects of antibiotics must be considered before their administration in a prophylactic regimen. However, follow up studies in an improved emergency medicine background have not achieved positive effects with this strategy [192-194].
Three observational studies support the matching for the complete rhesus phenotype and blood group K (Wayne 1995 prostate cancer zytiga order discount penegra online, Pearlman 1994 mens health 2013 buy penegra 100mg with visa, Russel 1984) prostate cancer home remedies buy generic penegra 100 mg online. The degree of immunisation in these patients decreases as a result of selection of rhesus phenotype compatible and K negative blood man health picture penegra 100 mg with visa. A recent study a b has also shown that the blood groups Fy, Jk, S and s are also important (in order of a b importance). By selecting Fy, Jk, S en s negative erythrocytes respectively for patients who are negative for these antigens (in order of importance), the degree of immunisation can be b decreased significantly (Schonewille 2006). As the frequency of Jk neg (51%) is greater a b than Jk neg (8%) in patients with sickle cell anaemia, particularly Jk compatible transfusions are important for these patients in order to prevent immunisation. Extensive selection of blood negative for these antigens can result in far-reaching reduction of allo-immunisation (Schonewille 2006, Castro 2002, Tahhan 1994). The degree of immunisation in these patients decreases as a result of selection of rhesus phenotype Level 2 compatible and K negative blood. C Schonewille 2006 b For patients with sickle cell anaemia, the frequency of Jk neg (51%) is a b greater than Jk neg (8%). Therefore, Jk compatible transfusion is important for these patients in order to prevent immunisation. More Level 3 extensive selection of blood negative for these antigens can result in farreaching reduction of allo-immunisation. C Schonewille 2006 (B), Castro 2002, (C) Tahhan 1994 Other considerations the selection choice of the compatible units is partly determined by the antigen determinations performed and the availability of typed units in the blood bank. Rhesus phenotype, K and Fy compatible blood should be selected for (potentially) transfusion-dependent patients with sickle cell anaemia or thalassaemia. If possible, b it is also recommended to select Jk, S and s negative erythrocytes (in order of importance) for patients who are negative for these antigens. Preferably, matches for Kidd, Duffy and Ss antigens are also indicated (in order of importance), if the presence of alloantibodies cannot be ruled out. The antigens of the Rhesus, Kell, Kidd and Ss systems can usually be detected serologically with monoclonal reagents if the patient has not received a transfusion in the past three months. It is therefore recommended to select rhesus phenotype and K compatible blood for these patients (Fluit 1990, Novaretti 2001, Stiegler 2001, Schonewille 1999, Arriaga 1995). The transfusion reactions described in older publications only occurred with strong cold antibodies and/or deep (~15 �C) hypothermia. Strong cold antibodies can cause problems in the standard Blood Transfusion Guideline, 2011 91 91 compatibility tests and deep hypothermia is now only used in combination with specific interventions. There is no evidence in the literature to support pre-operative screening for cold antibodies at room temperature for all patients being exposed to mild hypothermia (~ 30 �C). D Hoffman 2002 There is no evidence in the literature to support pre-operative screening for cold antibodies at room temperature for all patients being exposed to mild hypothermia. Level 4 D Judd 2006 Other considerations the transfusion reactions described in the literature only occurred in the case of the presence of strong cold antibodies and/or in surgery involving deep hypothermia. Following consultation with the anaesthesiologist, it may be desirable in some cases to determine the frequency of the clinically relevant cold antibody. Transfusion reactions and intravascular platelet degradation can occur at very high anti-A and/or anti-B titres and in the presence of haemolysins in the recipient. This is usually associated with slight haemolysis, although in an estimated 1 in 9000 patients minor incompatible platelet transfusions can cause severe � even fatal � haemolysis and renal failure (Mair 1998, Larsson 2000, Lozano 2003, Harris 2007). The amount of incompatible plasma can be reduced by 30% by using platelet storage solutions, or 95% of the plasma can be removed by hyper-concentration. Even this can be insufficient in the case of high anti-A and/or anti-B titres (Valbonesi 2000). It is advisable to avoid incompatible plasma for patients receiving multiple transfusions simultaneously or for children for whom the transfusion volume is 10 mL/kg body weight of the recipient. If this is not possible, the plasma should contain a relatively low antibody titre. In the Netherlands, it has been decided on practical and theoretical grounds to set a titre smaller than 128. The anti-A, anti-B titre determination is difficult to standardise (Harris 2007, Aubuchon 2008). There are no (inter)national guidelines for the quantity of incompatible plasma that may be administered with platelets. This policy can vary greatly, from warning the treating physician to volume reduction of platelets (Fung 2007). This is usually associated with slight haemolysis, but in approximately 1 in 9000 patients minor incompatible platelet Level 3 transfusions can cause severe � even fatal � haemolysis and renal failure. B Mair 1998 C Larsson 2000, Lozano 2003, Harris 2007 the transfusion of platelets in incompatible plasma can be largely reduced by the use of platelet storage solutions or by the removal of plasma. There are indications that these measures may be inadequate for very high antiLevel 3 A and/or anti-B titres. The minimum amount of erythrocytes capable of causing primary RhD immunisation is 0. The risk of RhD immunisation in patients who are immune suppressed is between 0 and 19% (Goldfinger 1971, Lozano 2003, Atoyebi 2000). This is an underestimate as antibodies can be demonstrated long after transfusion, on average 184 days (45 � 450 days). Unlike RhD, irregular erythrocyte antibodies due to platelet transfusions are rare, but cases have been described. Blood Transfusion Guideline, 2011 95 95 Platelet transfusions should preferably be RhD compatible. Level 3 C Mollison 1997, Cid 2005 Anti-RhD antibodies are found in 0 � 19% of immune suppressed patients. The actual immunisation frequency is probably higher because anti-RhD antibodies can only be demonstrated long after transfusion. Female RhD negative patients under the age of 45 years should only receive RhD negative platelet concentrates. As the apheresis plasma in the Netherlands is prepared using a method in which the the remaining 8 erythrocyte number is less than 1 x 10 /unit, the RhD blood group does not have to be taken into consideration. All donors are tested for irregular antibodies and are negative or have a titre lower than 32. Level 3 C Benjamin 1999 B Shanwell 2009 Other considerations European legislation means that it is compulsory for the RhD blood group to be stated on the label of the plasma component. For plasma transfusion, it is not necessary to take into consideration the RhD blood group. Prospective evaluation of a transfusion policy of D+ red blood cells into Dpatients. A review of the clinical effectiveness of routine antenatal anti-D prophylaxis for rhesus-negative women who are pregnant. Hemovigilance network in France: organization and analysis of immediate transfusion incident reports from 1994 to 1998. Comparing near misses with actual mistransfusion events: a more accurate reflection of transfusion errors. Comparison of the performance of four microtube column agglutination systems in the detection of red cell alloantibodies. A prospective study of the incidence of red cell alloimmunisation following transfusion. A prospective study to determine the safety of omitting the antiglobulin crossmatch from pretransfusion testing. The risk of an overt hemolytic transfusion reaction following the use of an immediate spin crossmatch. E et al Do patients with autoantibodies or clinically insignificant alloantibodies require anindirect antiglobulintest crossmatchfi Red blood cell alloantibodies after transfusion: factors influencing incidence and specificity. Evaluation of methods for detecting alloantibodies underlying warm autoantibodies. Coexistence of autoantibodies and alloantibodies to red blood cells due to blood transfusion.
These may include Herpes simplex-Injeel prostate cancer 8 gleason discount penegra 50mg online, Herpes zoster-Injeel define androgen hormone order online penegra, Hepatitis-Injeel mens health and fitness magazine order generic penegra pills, Grippe-Injeel prostate oncology associates buy 100 mg penegra free shipping, Fluor albus-Injeel, Trichomonaden-Fluor-Injeel, Vaccininum-Injeel, Variolinum Injeel, Diphtherinum-Injeel, Psorinum-Injeel, Tuberculinum-Injeel, etc. The Nosode should be administered in accordance with the principles of regressive vicariation. Claudication, intermittent (Charot�s disease) (Haemodermal impregnation phase) Aesculus compositum (main remedy) 3-6 times daily 10 drops, possibly in place of this Arteria-Heel Arnica-Heel Aesculus-Heel ana 8-10 drops 3 times daily Traumeel S tablets (regeneration of the sulphide enzymes) Traumeel S ointment to be rubbed in. Placenta suis-Injeel, Arteria suis-Injeel, possibly also Vena suis-Injeel as mixed injection i. See also endarteritis obliterans, disturbance of circulation, peripheral, thrombo-angitis obliterans, etc. Colic (Reaction or impregnation phases of various kinds, mainly entodermal) Spascupreel in frequent doses (tablets, suppositories, ampoules) or Atropinum compositum S suppositories, alternating with the other biotherapeutics and antihomotoxic agents such as Chelidonium-Homaccord (liver, gall bladder) Nux vomica-Homaccord (stomach, intestines) Veratrum-Homaccord (intestines, diarrhoea, dysentery) Reneel and Berberis-Homaccord (kidneys) Injection therapy Atropinum compositum is the principal remedy, otherwise the above Homaccords with Magnesium phosphoricum-Injeel (forte) for dysmenorrhoea, writer�s cramp and general convulsive disorders Erigotheel (stomach) Atropinum sulfuricum-Injeel forte (contains 0. At intervals also (for chronic colic-like conditions) Coenzyme compositum or the collective pack of Catalysts of the citric acid cycle. Colitis (Entodermal impregnation or reaction phase) (Main remedy: Nux vomica-Homaccord) Nux vomica-Homaccord at 8 a. Podophyllum compositum (colitis, haemorrhoids) Ignatia-Homaccord or Nervoheel (psychosomatic components) CinnamomumHomaccord S (discharge of blood) Cruroheel S (alternating remedy) Diarrheel S (dyspepsia, meteorism, diarrhoea) Traumeel S tablets (anti-inflammatory, antisuppurative action) Injection therapy Podophyllum compositum Nux vomica-Homaccord, Veratrum-Homaccord, Traumeel S, Galium-Heel and Colon suis-Injeel, possibly also Glandula suprarenalis suis-Injeel for the progressive autosanguis therapy, possibly repeated several times. Ignatia-Homaccord (in substitution for Nux vomica-Homaccord) Bacterium-coli-Injeel (forte), Bacterium proteus-Injeel (forte), Salmonella typhi-Injeel (forte) and Salmonella paratyphi B-Injeel (forte) for nosode therapy Mercurius praecipitatus ruber-Injeel (forte S) as intermediate remedy, Mucosa compositum (constitutional therapy, after-treatment) once to twice weekly i. Collagen-vascular diseases (Mainly mesenchymal impregnation or degeneration phases) the connective tissue which, with its high content of gelatinous substance (collagen), acts as sustentacular organ and, on the other hand, also as a spacious storehouse for numerous homotoxins which, at the moment, are not utilizable or intended for elimination, and in which both defensive reactions and the subsequent inflammatory processes take place, is subject to numerous possibilities of damage, particularly in the form of retoxic impregnation due to the inhibition of inflammatory and elimination processes of which, partly directly and partly indirectly, collagen diseases are the consequence (lupus erythematodes, etc. In this connection, the visible phenomenon can occur on the ectoderm and in addition, disturbances of the functions of the other organs (liver, etc. The principal remedies are Galium-Heel, Psorinoheel, Lymphomyosot, Hepeel; further, the progressive auto-sanguis therapy with Funiculus umbilicalis suis-Injeel, Hepar suisInjeel, Cutis suis Injeel and appropriate Injeels. Traumeel S tablets possibly in alternation with Aesculus compositum and Cruroheel S always interposed. Injection therapy Apart from Galium-Heel, Psorinoheel, Lymphomyosot, Hepeel, Traumeel S, Engystol N and Gripp-Heel, in particular also Echinacea compositum (forte) S, Thyreoidea compositum, Tonsilla compositum, Hepar compositum, Cutis compositum, for general revitalization, also Testis compositum (men) and Ovarium compositum (women), Coenzyme compositum, Ubichinon compositum (enzyme regeneration), in serious cases also Glyoxal compositum (a single injection, awaiting the subsequent effect), as well as Cutis compositum at intervals. Collapse (Haemodermal impregnation or degeneration phase) Aurumheel N drops, every 5-10 min. Injection therapy Carbo compositum, Traumeel S with Carbo vegetabilis-Injeel, Veratrum-Injeel forte S i. Medorrhinum-Injeel for cold sweat, the patient must remain uncovered (as for Carbo vegetabilis-Injeel) Vertigoheel with Traumeel S for brain injuries as mixed injection daily, in more serious cases twice daily i. Concussion (Neurodermal impregnation phase) Traumeel S drops in one to two-hourly alternation with Vertigoheel drops Injection therapy Traumeel S and Vertigoheel daily in the first few days, later 2-3 times weekly as mixed injection i. Hypericum-Injeel (forte) for brain and nerve injuries Cerebrum suis-Injeel for after-treatment once weekly, possibly alternating with Cerebrum compositum i. Condylomas (Ectodermal deposition phases) Psorinoheel 3 times daily 8-10 drops Schwef-Heel (intermediate remedy) Galium-Heel (detoxicating agent acting on the mesenchyme in neoplasm). Conjunctivitis (Ectodermal reaction phase) Oculoheel 1 tablet 3 times daily Belladonna-Homaccord for severe inflammation, as additional remedy Mercurius-Heel S for suppurative inflammation Apis-Homaccord for oedematous swelling Traumeel S tablets (anti-inflammatory, antisuppurative action) Traumeel S ointment externally Injection therapy Belladonna-Homaccord and Traumeel S, possibly also Euphrasia-Injeel and Aethiops antimonialis-Injeel as mixed injection or alternating i. Crocus-Injeel (forte) for burning of the eyes after reading for a short time, myiodesopsia Croton tiglium-Injeel for epiphora and photophobia, Euphrasia-Injeel (forte) for heliophobia by day, chemism Juglans-Injeel for chronic gonorrhoeal ophthalmia Ruta-Injeel (forte) after overstrain Mucosa compositum (remedy for affections of the mucous membranes) interposed once weekly i. Constipation (Entodermal deposition phase) (Main remedy: Nux vomica-Homaccord) Nux vomica-Homaccord 8-10 drops at 8 a. Hepeel for meteorism, 1 tablet, several times Gastricumeel for swelling in the epigastrium, 1 tablet, several times Injection therapy Hepeel (or Injeel-Chol or Chelidonium-Homaccord) alternating with Nux vomicaHomaccord, possibly Erigotheel and Traumeel S i. Nux vomica-Injeel (forte) S in typical cases (futile tenesmus) Asthma-Nosode-Injeel for chronic, spasmodic constipation, Bacterium coli-Injeel interposed Collective pack of the Catalysts of the citric acid cycle or also Mucosa compositum (remedy for affections of the mucous membranes), possibly also Hepar compositum (therapeutic agent for affections of the hepatic functions) once to twice weekly i. Corneal opacity (Ectodermal deposition or impregnation phase) (Main remedy: Galium-Heel) Galium-Heel at 8 a. Injection therapy Calcium fluoratum-Injeel, Galium-Heel, Psorinoheel and possibly Engystol N alternating or mixed i. Corns (Ectodermal deposition phase) Arsuraneel 1 tablet 3 times daily Antimonium crudum-Injeel (forte), as ampoules to be taken orally. Mercurius bijodatus-Injeel (forte S) for painful conditions, Arsenicum album-Injeel (forte) S (keratinization), Cutis compositum (action on the skin), also Cutis suis Injeel in chronic cases and relapses. Traumeel S ointment, possibly Calendula-Salbe-Heel S ointment or Paeonia-SalbeHeel ointment. Coronary diseases See angina pectoris, myocardial infarction (heart attack), disturbance of circulation, coronary insufficiency, neurocirculatory asthenia, atrial fibrillation. Coronary insufficiency (Haemodermal impregnation or deposition phase) Cactus compositum S 3 times daily 10 drops, or Cardiacum-Heel at 8 a. Aurumheel N drops for myocardial weakness substituted for Cralonin, GlonoinHomaccord N drops for anginal attacks Traumeel S tablets (regeneration of the sulphide enzymes) Gastricumeel for gastrocardial syndrome (possibly with Spascupreel, Duodenoheel and Bryaconeel, allowing them to be taken simultaneously several times daily). Coughs See bronchitis, laryngitis, pertussis, pleurisy, pneumonia, influenza, etc. Cradle cap (seborrhoea) (Ectodermal reaction phase) (Main remedy: Graphites-Homaccord) Graphites-Homaccord at 8 a. Abropernol, Dulcamara-Homaccord and Mezereum-Homaccord possibly as intermediate remedies. Traumeel S ointment to be applied daily (possibly with the head covered by a hood). Cramp See colic, spasms, epilepsy, nephrolithiasis, cholangitis, torticollis spasticus, gastroenteritis etc. Croupous coughs (Entodermal reaction or impregnation phase) (Main remedy: Strumeel forte N; spongioid action) Strumeel forte N 8-10 drops in the morning Aconitum-Homaccord 8-10 drops at midday Husteel 8-10 drops in the evening In the case of an attack, all 3 remedies in frequent doses (every 3-5 min. Injection therapy Aconitum-Homaccord with Spongia-Injeel (forte) and Hepar sulfuris-Injeel (forte), possibly Traumeel S i. Mucosa compositum (in chronic cases: remedy for affections of the mucous membranes) Cushing�s syndrome (Ectodermal or mesenchymal degeneration phase) (Main remedy: Hormeel S) Hormeel S at 8 a. Aesculus compositum (regulation of the peripheral circulation), possibly daily, alternating with the above preparations. Injection therapy the above preparations (in ampoule form) alternating or mixed i. Thyreoidea compositum (stimulating effect on the glandular and connective tissue functions) and Placenta compositum (regulation of the peripheral circulation) with Testis compositum (for men) or Ovarium compositum (for women) interpolated in alternation. Grippe-Nosode-Injeel (forte), Diphtherinum-Injeel (forte), Sinusitis-NosodeInjeel (forte) and Staphylococcus-Injeel (forte) and Glandula thyreoidea suis-Injeel (forte) interpolated at intervals, possibly with Engystol N, Traumeel S, etc. Coenzyme compositum, possibly also Ubichinon compositum, possibly also the collective pack of Catalysts of the citric acid cycle to be interpolated to stimulate the enzyme functions. Cysts (According to localization, diverse deposition phases) (Main remedy: Apis-Homaccord) Apis-Homaccord at 8 a. Traumeel S tablets (resorptive mercury and enzyme regenerating sulphide effect) Aesculus compositum (regulation of the peripheral circulation) Injection therapy Apis-Homaccord, Galium-Heel and Psorinoheel alternating i. Cantharis compositum S (diseases of the kidneys and bladder) Solidago compositum S (constitutional therapy, chronic cystalgia) Coxsackie-Virus-A9 or B4-Injeel (forte) is often specifically effective. Sarsaparilla-Injeel as intermediate remedy, Urinum gravidarum D 200 in extremely chronic cases, Medorrhinum-Injeel (forte) and Tuberculinum-Injeel (forte) as intermediate remedy (nosodes). Bacterium coli-Injeel, possibly also Otitis media-Nosode-Injeel, Myrtillus-Injeel (forte). Mucosa compositum (therapeutic agent for affections of the mucous membrane) and Echinacea compositum (forte) S (general remedy for inflammation) as intermediate injections, otherwise Vesica urinaria suis-Injeel in chronic cases. Traumeel S tablets (anti-inflammatory, antisuppurative action) Cruroheel S (fistular suppurations) Psorinoheel (chronicity) Traumeel S ointment externally Belladonna-Homaccord in incipient cases Injection therapy Belladonna-Homaccord and Traumeel S alternating or mixed i. Pulsatilla-Injeel (forte) S as auxiliary remedy Mucosa compositum (chronic affections of the mucous membranes). Decubitus ulcers (Ectodermal, possibly mesenchymal reaction or degeneration phase) (Main remedy: Traumeel S) Traumeel S-liquid 8-10 drops at 8 a. Aesculus compositum (promotion of the circulation) possibly administered in daily alternation. Arnica-Heel possibly in place of Traumeel S Traumeel S ointment dressings, Kamillen-Salbe-Heel S ointment Injection therapy Traumeel S i. Dedifferentiation Phases (formerly Neoplasm Phases) Delirium tremens (Neurodermal impregnation or degeneration phase) Veratrum-Homaccord initially 1/4 to 1/2 hourly, later hourly to two-hourly, alternating with Psorinoheel. Gelsemium-Homaccord and Spascupreel as auxiliary remedy Injection therapy the oral preparations also per injection (ampoule form), in addition Hyoscyamus-Injeel (forte) for Korsakoff�s psychosis Stramonium-Injeel (forte) for muttering delirium Bacterium proteus-Injeel (forte), possibly also Medorrhinum-Injeel (forte) for nosode therapy Cerebrum compositum (intermediate injections and after-treatment) once weekly i.
Lab and hospital staff benefit from faster and more reliable data capture and improved information access prostate cancer mayo clinic discount penegra 50mg on-line. At the hospital mens health zero excuses workout buy generic penegra from india, kit inventory is managed to prostate cancer under 40 order penegra uk ensure kits are available and not expired prostate cancer metastasis buy on line penegra. The system is integrated with the state courier system to make placing courier orders even easier. To reduce delays and avoid lost envelopes, the system notifies the hospital and lab if specimen delivery is late. Once specimens have been received by the lab, the system automatically notifies the hospital, meeting accreditation requirements. This process and the the process of working with other programs to better understand how others are handling similar challenges has led to insights in several different areas. However, there are differences in how "quality data" is defined, the information that is requested on the collection card, and how the data is entered and validated. The terms "case management" and "follow up" are often used to refer to the same set of activities whereas the activities themselves might actually benefit from a stricter interpretation of these terms. Results: the results of the analytical performance and the comparison with the archived sample set will be presented in the poster. No major interference effects by relevant endogenous or exogenous compounds are expected. Presenter: Petra Furu, Global Business Manager, PerkinElmer, Turku, Finland, Phone: 358. In this development study, typical analytical performances of multiple newly added markers were studied and demonstrated. All the samples were tested with the developed NeoBase2 non-derivatized assay protocol. After the extraction step, the sample aliquots (100 �L/well) were transferred to the new microplate. Results: Study results to be presented suggest all the investigated new markers can be measured with good linearity over adequately wide measuring ranges covering the expected normal and abnormal clinically relevant concentration areas. Based on the recent literature, abnormal profiles of one or more of these new markers may be indicative for. The objectives of this study were to further demonstrate the benefits of new streamlined NeoBase2 assay, and provide typical analytical performance comparison between these two non-derivatized assays. All the study samples were tested with the both compared assay protocols in order to study their between method correlation. After the extraction step, the sample aliquots were transferred to the new microplate. Results and Conclusions: Study results to be presented suggest the compared assays show relatively good overall correlation with majority of studied analytes including multiple amino acids, acylcarnitines and succinylacetone. Mainly due to several procedural improvements implemented in the novel NeoBase2 assay. This means all the screening laboratories aiming to update later their current method to the new NeoBase2 method should run some representative parallel assay transition period prior to the assay change and update their currently applied reference ranges accordingly if needed. We also show that a simple wash buffer improves the assay performance as compared to the commonly used Qiagen S2 buffer. With more options available than most laboratories have resources, there is a demand to make testing even more efficient than previously required. No longer is simply decreasing the cost per result sufficient, the real estate on a plate favors multiplex assays over singleplex tests. Furthermore, automation of the assay can significantly decrease the amount of hands-on time needed from the staff. Continuing with assay-efficiency, this work will present how to perform both assays using the Zephyr automated liquid handler by PerkinElmer. A comparison between the manual and automated processes will show that there is no decrease in the assays� precision using automation and a time-savings analysis will demonstrate the improved workflow that one can expect from such a system. All samples, except the 2016controls were stored at -20� C with desiccant until use. Measurements of the C20-C26 acylcarnitines were close to the limit of quantification and seem less useful for screening purposes. Presenter: Peter Schielen, Head, Reference Laboratory, Neonatal Screening, Bilthoven, Utrecht, the Netherlands, Email: peter. Mansour2; 1Saint Joseph University, Beirut, Lebanon, 2University of Balamand, Balamand, Lebanon Background and aims: With the high rate of consanguineous marriages in Lebanon, the risk of occurrence of inborn errors of metabolism seems to be higher than expected. In the absence of national registry for rare disorders and national newborn screening programs, community based universities established newborn screening laboratories and metabolic units follow up. Currently, 60 per cent of Lebanese newborns benefit from expanded out-of-pocket newborn screening programs. Accurate statistical number remain far from our grasp especially that many patients remain undiagnosed and fail to reach appropriate medical care. Methods: We present here an overview of the newly diagnosed patients with inborn errors of metabolism since the expansion of the newborn screening panel by tandem mass spectrometry in late 2006. Results: 107 new patients with confirmed metabolic disorders were diagnosed among 202 000 babies screened between November 2006 and March 2017. The use of Ratio�s was imminent for adequate diagnosis as the relative metabolite may not be elevated at time of sampling like C3/C2 and C3/C16. Conclusions: the high incidence of metabolic diseases found justify the screening that should mandated for all newborns integrated in the national health policy in Lebanon. Families and group advocate are gathered and an awareness video is under preparation to highlight that it is time for policy makers to consider it. In addition, many improvement had been made in the management of such specific disorders that may faces with the lack of pharmacological resources and the limited access to specific diet formulas. Presenter: Issam Khneisser, Unit Head, Newborn Screening Laboratory, Medical Genetic Unit, Saint Joseph University, Beirut, Lebanon, Email: issam. Products from multiple vendors were compared for panel variant content, workflow, cost, and future flexibility. After consideration of these factors, preliminary experiments were begun to assess potential implementation of the ThermoFisher Scientific genotyping platform. Phe508del), is located within one of these regions and can require concurrent detection of c. Additionally, there are data management and information technology issues to address for workflow. Additionally, this platform could be leveraged for genotyping other newborn screening disorders. The median and 99th centile for the sum of glutamine and lysine were 212 and 958 uM, respectively. Median and 99th centile were: 65 and 534 uM (lysine), and 507 and 899 uM (glutamine). Presenter: Ralph Fingerhut, Swiss Newborn Screening Lab, University Children�s Hospital, Zurich, Switzerland, Phone: 41. Methodology: In 2016, a survey was distributed to gain stakeholder information on specimen collection times, shipping methods, and opinions on what would be helpful for improvement. The responses from the survey allowed for identification of barriers that birthing facilities are facing that could be addressed by the Newborn Screening Program. These barriers were addressed in the development of various education/training materials. Materials developed include: an interactive online component, a print component, and an in-person component. A post evaluation for timeliness improvements will be completed during the months following implementation of the education materials. Conclusion: Further improvements are expected after the implementation of training and educational opportunities for providers and stakeholders in facilities across the state. Our objective during this cycle is to identify barriers to timeliness with subsequent implementation of initiatives to improve time to diagnosis. Methodology: An evaluation of our current laboratory (pre-analytical, analytical, and post analytical) and follow-up processes were conducted to identify issues and potential solutions for improvement. Therefore, any specimens requiring confirmation of initial results from the last run of the week are postponed for testing until the next week resulting in delayed final reporting. Conclusion: Initiatives to address these issues are a work in progress and we anticipate an postimplementation evaluation to determine the impact any changes have made to our timeliness project. If a baby has been identified as having an abnormal first tier analyte and then is found to have two molecular variants that might predict a later onset of disease is this a case for newborn screeningfi Follow up programs of different states are considering this information differently, leading to nonconformity in detection numbers and overall prevalence.
Buy 50 mg penegra amex. Men's Health PITCH: Mediterranean Diet.