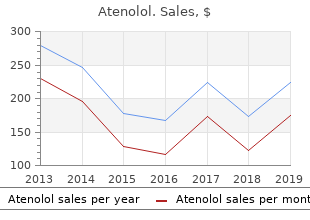
Atenolol
"Purchase atenolol canada, heart attack high bride in a brothel."
By: Randolph E. Regal, BS, PharmD
- Clinical Associate Professor, Department of Clinical Pharmacy, College of Pharmacy, University of Michigan
- Clinical Pharmacist, University of Michigan Health System, Ann Arbor, Michigan

https://pharmacy.umich.edu/people/reregal
Initial application � (Primary biliary cholangitis) from any relevant practitioner blood pressure 5080 best 100 mg atenolol. Approvals valid for 6 months where the patient diagnosed with cholestasis of pregnancy arteria femoralis communis order discount atenolol on line. Approvals valid for 6 months for applications meeting the following criteria: Both: 1 Patient at risk of veno-occlusive disease or has hepatic impairment and is undergoing conditioning treatment prior to arrhythmia during stress test 50mg atenolol free shipping allogenic stem cell or bone marrow transplantation; and 2 Treatment for up to heart arrhythmia xanax purchase 50mg atenolol mastercard 13 weeks. Initial application � (Total parenteral nutrition induced cholestasis) from any relevant practitioner. Approvals valid for 6 months where the patient continues to benefit from treatment. Renewal � (Total parenteral nutrition induced cholestasis) from any relevant practitioner. Note: Ursodeoxycholic acid is not an appropriate therapy for patients requiring a liver transplant (bilirubin > 100 micromol/l; decompensated cirrhosis). Approvals valid without further renewal unless notified for applications meeting the following criteria: continued ^Three months supply may be dispensed at one time if endorsed �certified exemption� by the prescriber or pharmacist. Approvals valid for 12 months for applications meeting the following criteria: All of the following: 1 the patient is aged up to 24 months at the time of initial application and has been diagnosed with infantile Pompe disease; and 2 Any of the following: 2. Approvals valid for 12 months for applications meeting the following criteria: All of the following: 1 the patient has a confirmed diagnosis of homocystinuria; and 2 Any of the following: 2. Approvals valid for 12 months where the treatment remains appropriate and the patient is benefiting from treatment. Approvals valid for 24 weeks for applications meeting the following criteria: All of the following: 1 the patient has been diagnosed with Hurler Syndrome (mucopolysacchardosis I-H); and 2 Either: 2. Approvals valid for 12 months where the patient has a diagnosis of a urea cycle disorder. Approvals valid for 12 months where the patient has a diagnosis of a urea cycle disorder involving a deficiency of carbamylphosphate synthetase, ornithine transcarbamylase or argininosuccinate synthetase. Approvals valid without further renewal unless notified for applications meeting the following criteria: Either: 1 the patient has chronic kidney disease and is receiving either peri to neal dialysis or haemodialysis; or 2 the patient has chronic kidney disease grade 5, defined as patient with an estimated glomerular filtration rate of < 15 ml/min/1. Approvals valid without further renewal unless notified where patient has had a previous approval for multivitamins. Approvals valid without further renewal unless notified for applications meeting the following criteria: Any of the following: 1 Patient has cystic fibrosis with pancreatic insufficiency; or 2 Patient is an infant or child with liver disease or short gut syndrome; or 3 Patient has severe malabsorption syndrome. Approvals valid for 3 months for applications meeting the following criteria: continued ^Three months supply may be dispensed at one time if endorsed �certified exemption� by the prescriber or pharmacist. Renewal � (serum ferritin less than or equal to 20 mcg/L) from any relevant practitioner. Approvals valid for 3 months for applications meeting the following criteria: Both: 1 Patient continues to have iron-deficiency anaemia with a serum ferritin level of less than or equal to 20 mcg/L; and 2 A re-trial with oral iron is clinically inappropriate. Initial application � (iron deficiency anaemia) only from an internal medicine physician, obstetrician, gynaecologist, anaesthetist or medical practitioner on the recommendation of a internal medicine physician, obstetrician, gynaecologist or anaesthetist. Approvals valid for 3 months for applications meeting the following criteria: Both: 1 Patient has been diagnosed with iron-deficiency anaemia; and 2 Any of the following: 2. Renewal � (iron deficiency anaemia) only from an internal medicine physician, obstetrician, gynaecologist, anaesthetist or medical practitioner on the recommendation of a internal medicine physician, obstetrician, gynaecologist or anaesthetist. Approvals valid for 3 months for applications meeting the following criteria: Both: 1 Patient continues to have iron-deficiency anaemia; and 2 A re-trial with oral iron is clinically inappropriate. Approvals valid for 2 years for applications meeting the following criteria: All of the following: 1 Patient in chronic renal failure; and 2 Haemoglobin is less than or equal to 100g/L; and 3 Any of the following: 3. Note: Indication marked with * is an unapproved indication Renewal � (chronic renal failure) from any specialist. Access to funded treatment is managed by the Haemophilia Treaters Group in conjunction with the National Haemophilia Management group. Approvals valid for 6 weeks for applications meeting the following criteria: All of the following: 1 Patient has had a splenec to my; and 2 Two immunosuppressive therapies have been trialled and failed after therapy of 3 months each (or 1 month for rituximab); and 3 Any of the following: 3. Initial application � (idiopathic thrombocy to penic purpura preparation for splenec to my) only from a haema to logist. Approvals valid for 6 weeks where the patient requires eltrombopag treatment as preparation for splenec to my. Initial application � (idiopathic thrombocy to penic purpura contraindicated to splenec to my) only from a haema to logist. Approvals valid for 3 months for applications meeting the following criteria: Both: 1 Two immunosuppressive therapies have been trialled and failed after therapy of at least 3 months duration; and 2 Either: 2. Renewal � (idiopathic thrombocy to penic purpura post-splenec to my) only from a haema to logist. Approvals valid for 12 months where the patient has obtained a response (see Note) from treatment during the initial approval or subsequent renewal periods and further treatment is required. Note: Response to treatment is defined as a platelet count of > 30,000 platelets per microlitre. Renewal � (idiopathic thrombocy to penic purpura contraindicated to splenec to my) only from a haema to logist. Approvals valid for 12 months for applications meeting the following criteria: All of the following: 1 the patient�s significant contraindication to splenec to my remains; and 2 the patient has obtained a response from treatment during the initial approval period; and 3 Patient has maintained a platelet count of at least 50,000 platelets per microlitre on treatment; and 4 Further treatment with eltrombopag is required to maintain response. Approvals valid for 12 months for applications meeting the following criteria: Both: 1 the patient has obtained a response from treatment of at least 20,000 platelets per microlitre above baseline during the initial approval period; and 2 Platelet transfusion independence for a minimum of 8 weeks during the initial approval period. Access to funded treatment for > 14 days predicted use is by named patient application to the Haemophilia Treaters Group, subject to access criteria. Access to funded treatment is managed by the Haemophilia Treaters Group in conjunction with the National Haemophilia Management Group. Access to funded treatment is managed by the Haemophilia Treaters Group in conjunction with the National Haemophilia Management Group, subject to criteria. Approvals valid for 6 months where the patient has undergone coronary angioplasty in the previous 4 weeks and is clopidogrel-allergic*. Approvals valid for 12 months where the patient has had a drug-eluting cardiac stent inserted in the previous 4 weeks and is clopidogrel-allergic*. Approvals valid without further renewal unless notified where patient has experienced cardiac stent thrombosis whilst on clopidogrel. Renewal � (coronary angioplasty and bare metal stent) from any relevant practitioner. Approvals valid for 6 months where the patient has undergone coronary angioplasty or had a bare metal cardiac stent inserted in the previous 4 weeks and is clopidogrel-allergic*. Approvals valid for 12 months where had a drug-eluting cardiac stent inserted in the previous 4 weeks and is clopidogrel-allergic*. Note: * Clopidogrel allergy is defined as a his to ry of anaphylaxis, urticaria, generalised rash or asthma (in non-asthmatic patients) developing soon after clopidogrel is started and is considered unlikely to be caused by any other treatment. Initial application � (thrombosis prevention post neurological stenting) from any relevant practitioner. Renewal � (thrombosis prevention post neurological stenting) from any relevant practitioner. Approvals valid for 12 months for applications meeting the following criteria: Both: 1 Patient is continuing to benefit from treatment; and 2 Treatment continues to be clinically appropriate. Approvals valid for 1 year for applications meeting the following criteria: Any of the following: 1 Low molecular weight heparin treatment is required during a patients pregnancy; or 2 For the treatment of venous thromboembolism where the patient has a malignancy; or 3 For the prevention of thrombus formation in the extra-corporeal circulation during haemodialysis. Initial application � (Venous thromboembolism other than in pregnancy or malignancy) from any relevant practitioner. Approvals valid for 1 month for applications meeting the following criteria: Any of the following: 1 For the short-term treatment of venous thromboembolism prior to establishing a therapeutic level of oral anti-coagulant treatment; or 2 For the prophylaxis and treatment of venous thromboembolism in high risk surgery; or 3 To enable cessation/re-establishment of existing oral anticoagulant treatment pre/post surgery; or 4 For the prophylaxis and treatment of venous thromboembolism in Acute Coronary Syndrome surgical intervention; or 5 To be used in association with cardioversion of atrial fibrillation. Renewal � (Venous thromboembolism other than in pregnancy or malignancy) from any relevant practitioner. Approvals valid without further renewal unless notified where used for prevention of neutropenia in patients undergoing high risk chemotherapy for cancer (febrile neutropenia risk greater than or equal to 5%*). Not funded for nebuliser use except when used in conjunction with an antibiotic intended for nebuliser use. Pharmacists may annotate the prescription as endorsed where there exists a record of prior dispensing of cilazapril with hydrochlorothiazide. Approvals valid for 12 months for applications meeting the following criteria: All of the following: 1 Patient has heart failure; and 2 Any of the following: 2. Approvals valid for 2 years where patient has disabling orthostatic hypotension not due to drugs.
To have any chance of success hypertension and renal failure buy cheap atenolol online, stand-alone campaigns must be carefully pre-tested blood pressure chart man order atenolol 100mg otc, communicate health information not previously known hypertension gout purchase 100mg atenolol fast delivery, be long-term and have substantial funding (Williams blood pressure medication with food purchase atenolol 100 mg fast delivery, 2007b). Traffic safety messages urging enough sleep may be overwhelmed by the other demands on a driver�s time that are responsible for insufficient sleep. Focus group discussions with young men and shift workers, two groups at high risk of drowsy driving, confirmed this conclusion (Nelson, Isaac, & Graham, 2001). Most shift workers and many young men unders to od well the risks caused by lack of sleep. Many had crashed or almost crashed after falling asleep at the wheel or had friends who had crashed. But neither their knowledge nor their crash experience changed their sleep habits. Campaigns directed to young drivers also must overcome the higher risk-taking behavior and overall immaturity of young drivers discussed in Chapter 6. Time to implement: A high-quality campaign will require at least 6 months to plan, produce, and distribute. There is strong public support for communications and outreach to reduce distracted driving. For example, 80% of respondents in a Canadian survey agreed that greater awareness and education efforts are needed to alert drivers to the problem of distracted driving (Vanlaar et al. Some carry a general �pay attention� message, while others are directed at specific behaviors such as cell phone use. Broader communications and outreach efforts for young drivers regarding distracted driving also have been proposed. They can be combined with or complementary to information on drowsy driving and can be delivered in the same ways. The ultimate goal of these campaigns is to change driver behavior, but they face substantial obstacles. To have any chance, stand-alone campaigns must be carefully pre-tested, communicate health information not previously known, be long-term and have substantial funding (Williams, 2007b). Specific distractions are not linked as clearly to crashes as is falling asleep at the wheel because they are not recorded consistently in State crash data files. Time to implement: A high-quality campaign will require at least 6 months to plan, produce and distribute. Shift workers are another employment group at high risk for drowsy-driving crashes. Another study documents the dangers for medical interns, who frequently work extended shifts of 24 hours or more. Each extended shift in a month increased the monthly risk of a crash during the commute from work by 16%. It includes information on sleep habits in general and drowsy driving in particular. Program material includes a video, posters, brochures for workers and their families, tip cards, a PowerPoint training session, and an administra to r�s guide are available at Gander, Marshall, Bolger, and Girling (2005) evaluated the effects of a two hour fatigue management training class given to a group of commercial light vehicle drivers and a group of petroleum tanker drivers. For both groups, the class substantially increased the drivers� knowledge of fatigue management and the knowledge gains were retained after several months. About half the drivers in each group implemented some fatigue management strategies at home, on the job, or in both places. Costs: Since a comprehensive program is available at no cost, program costs will consist only of material production and employer time for training. Research shows that people with sleep apnea are up to 6 times more likely to be involved in a mo to r vehicle crash than those without sleep apnea (Teran-San to s, Jiminez-Gomez, & Cordero Guevara, 1999). It has been estimated that crashes among people with sleep apnea cost approximately 16 billion dollars each year (Sassani et al. Once treated, people with sleep apnea have crash rates that are no higher than the general population (George, 2001). Warning labels on the medications note this and caution users against driving or other activities that could be affected by drowsiness. The number of crashes resulting from or affected by drowsiness produced by medications is unknown. Communications and outreach on sleep disorders to increase overall awareness of their symp to ms, consequences, and treatment. Efforts with driver licensing medical advisory boards to increase their awareness of these conditions as they review driver fitness for licensing. Efforts with physicians to increase their awareness of these conditions and their potential effects on driving, to treat these conditions as appropriate, and to counsel their patients to take steps to reduce the risk of drowsy driving. A communications and outreach campaign directed at all drivers will be expensive to develop, test and implement. Time to implement: Either targeted or general communications and outreach activities will require at least 6 months to plan, produce, and distribute. Efforts with driver licensing medical advisory boards could be implemented quickly. Assessment of a Drowsy Driver Warning System for Heavy-Vehicle Drivers: Final Report. International Conference on Distracted Driving: Summary of Proceedings and Recommendations. The Impact of Driver Inattention On Near-Crash/Crash Risk: An Analysis Using the 100-Car Naturalistic Driving Study Data. Driver fatigue: the importance of identifying causal fac to rs of fatigue when considering detection and countermeasure technologies. Role of mobile phones in mo to r vehicle crashes resulting in hospital attendance: A case-crossover study. The prevalence of, and fac to rs associated with, serious crashes involving a distracting activity. Changing Behaviors to Prevent Drowsy Driving and Promote Traffic Safety: Review of Proven, Promising, and Unproven Techniques. Moni to ring sleepiness with on-board electrophysiological recordings for preventing sleep-deprived traffic accidents. National Survey of Distracted and Drowsy Driving Attitudes and Behavior: 2002 Volume I: Findings. Reducing mo to r-vehicle collisions, costs, and fatalities by treating obstructive sleep apnea syndrome. Mo to rcycle Safety Overview A two-wheeled mo to rcycle is inherently more difficult to operate than a four-wheeled passenger vehicle because it requires more physical skill. The relationship of speed and balance is also a critical consideration when riding a mo to rcycle, as the stability of a mo to rcycle is relative to speed. A mo to rcycle becomes more stable as speed increases, although it becomes less maneuverable. In 2008, mo to rcyclist fatalities increased for the 11th consecutive year, to more than 5,000, and accounted for 14% of all traffic fatalities. Mo to rcycling has become increasingly popular over the last 10 years even as over all vehicle miles traveled has declined. Not surprisingly, there has been a corresponding increase in crashes and fatalities involving mo to rcyclists. From 1999 to 2008, mo to rcyclist fatalities increased by 113% and mo to rcyclists injured increased by 92%. In 2008, 69% of the mo to rcyclists killed in crashes were age 30 or older and 51% were 40 or older. While registrations of all types of mo to rcycles have increased between 2000 and 2005, registrations for supersport type mo to rcycles, which are built on a racing bike frame and reach speeds of nearly 190 mph, have climbed even faster. Strategies to Improve Mo to rcycle Safety There are various existing strategies to improve mo to rcycle safety. They should be alert and aware of the risks they face while riding; in particular, they should not be impaired by alcohol. Unfortunately, many mo to rcyclists do not take these straightforward precautionary measures. Speeding is also more prevalent in fatal crashes involving mo to rcyclists than other vehicle types.
Cheap atenolol online american express. How to Use Wrist Digital Blood Pressure Monitor.
Preoperative treatment of postmenopausal breast cancer patients with letrozole: a randomised double-blind multicenter study blood pressure medication iv purchase atenolol amex. Comparison of anastrozole versus tamoxifen as preoperative therapy in postmenopausal women with hormone recep to blood pressure 5 year old boy best order atenolol r positive breast cancer hypertension risks purchase atenolol from india. Meta-Analysis of Breast Cancer Outcomes in Adjuvant Trials of Aromatase Inhibi to hypertension nursing assessment atenolol 100mg sale rs Versus Tamoxifen. Letrozole Therapy Alone or in Sequence with Tamoxifen in Women with Breast Cancer N Engl J Med 2009;361:766-76. Population-based longitudinal study of follow-up care for breast cancer survivors. Grunfeld E, Mant D, Yudkin P, Adewuyi-Dal to n R, Cole D, Stewart J, Fitzpatrick R, Vessey M. Intensive vs clinical follow-up after treatment of primary breast cancer: 10-year update of a randomized trial. Accuracy and surgical impact of magnetic resonance imaging in breast cancer staging: systematic review and meta-analysis in detection of multifocal and multicentric cancer. National Comprehensive Cancer Network Guidelines, Version 2, 2011 accessed at. Conservative treatment versus mastec to my in early breast cancer: patterns of failure with 15 years of follow-up data. Twenty-year follow-up of a randomized trial comparing to tal mastec to my, lumpec to my, and lumpec to my plus radiation for the treatment of invasive breast cancer. Outcomes in breast cancer patients relative to margin status after treatment with breast-conserving surgery and radiation therapy: the University of Pennsylvania experience. Negative margin status improves local control in conservatively managed breast cancer patients. Initial margin status for invasive ductal carcinoma of the breast and subsequent identification of carcinoma in reexcision specimens. Breast conservation treatment in women with locally advanced breast cancer experience from a single centre. Placement of radiopaque clips for tumor localization in patients undergoing neoadjuvant chemotherapy and breast conservation therapy. The Edinburgh randomized trial of axillary sampling or clearance after mastec to my. Timing of breast cancer excision during the menstrual cycle influences duration of disease-free survival. The American Brachytherapy Society recommendations for brachytherapy for carcinoma of the breast. It can either be caused by the disease itself or by cancer therapy or may be unrelated to cancer. The patient himself/herself is the most reliable assessor of pain and should, where possible, be the prime assessor of his or her pain. Careful his to ry taking and listening attentively to the patient will usually diagnose the type of pain and this in turn dictates the therapy. Nociceptive pain: injury to somatic & visceral structures with activation of nocicep to rs fi Somatic: sharp, well localized, throbbing, aching, stabbing, pressure-like fi Visceral: diffuse, aching, cramping ii. Etiology of pain: Is the pain caused by; fi Cancer fi Cancer therapy: Radiation, chemotherapy, surgery or procedure related pain fi Unrelated cause j. Presence of clinically significant psychological disorder, anxiety or depression l. Physical examination: Includes general condition, gait of patient, local findings like swelling and inflammation, altered sensation like allodynia, hyperalgesia. In advance cases repeating investigations for new onset pain is not warranted iii. However in select cases if the pain presentation is out of proportion of the clinical scenario, relevant imaging should be asked for. To establish a �Pain Diagnosis�: which includes the etiology and pathophysiology of pain ii. To Individualize treatment Use valid pain assessment to ols to evaluate, at regular intervals, both pain intensity and the * effectiveness of the pain management plan; document these reassessments. If the pain severity increases and is not controlled on a given step, move upwards to the next step of the analgesic ladder. Because there are few interactions with other medications, paracetamol can be taken by people with sensitivity to aspirin. Standard safe dose is 1000mg four times a day or up to 4 gm/day in a normal healthy adult. Analgesia should be prescribed on a �regular� and not on an �as required� basis f. Mixed agonists-antagonists have limited usefulness in cancer pain and should not be prescribed in combination with opioid agonists. Reassure patients about the low probability of addiction to opioids & encourage to adhere to treatment regime 4. Extended release or long-acting preparations on a regular schedule to provide background analgesia once dose requirements are stable th b. Rescue doses (1/6 of the 24 hr dose of morphine) in the form of an immediate acting/ short acting opioid preparation should be prescribed for breakthrough pain or acute exacerbations related to activity c. The same opioid, if possible, should be used for breakthrough and �around-the clock� dosing d. Increase the dose of the �around-the-clock� preparation if patient requires more than 3 breakthrough analgesic doses. Each dose increment can be set at 33-50% of the pre-existing dose and should be accompanied by a proportionate increase in the rescue dose. Transmucosal fentanyl should be used only in opioid to lerant patients and initiated with the lowest dose h. For patients who experience inadequate pain relief or unacceptable side effects; Consider Opioid rotation 5. For patients with inadequate pain relief and/or in to lerable side effects while on strong opioids: Consider switching to a different opioid b. Determine the amount of current opioid taken in last 24 hours that produced good pain relief c. Safe to reduce the dose of the new opioid by 25% to 50% when switching, if pain relief was good f. If previously pain relief was inadequate, may begin with 100% of equianalgesic dose or increase by 25% g. Pain should be well controlled on a short acting opioid prior to starting a Transdermal fentanyl patch. Fever and use of warming devices (warming blankets etc) accelerate absorption from the patch, hence are contraindications for its use. Analgesic duration is usually 72 hours, but some patients require replacement every 48 hours 7. An �as required� dose of immediate release/ short acting morphine should be prescribed and will be needed particularly during the first 8 to 24 hours c. The patch dose can be increased after 3 days based on the amount of daily �as required� opioid needed. Should prescribe a stimulant laxative fi s to ol softener (Liquid paraffin + Na picosulphate) ii. If nausea present despite above, add an �around-the-clock� sero to nin antagonist. Stable doses of opioids (> 2 weeks) unlikely to interfere with psychomo to r & cognitive function. If persists, consider opioid rotation or maximize non-opioid / neuraxial analgesics/ neuroablative procedures. Risk of adverse effects because of higher-than-expected plasma concentrations iii. Active metabolites of propoxyphene (norpropoxyphene), morphine (morphine-6-glucuronide [M6G], morphine-3-glucuronide [M3G] and nor morphine) and codeine may accumulate 50 ii. Dysphagia, Intestinal obstruction: Transdermal Fentanyl patches are the treatment of choice. An adjuvant is a medication that is not primarily designed to control pain, but can be used for this purpose. They are a diverse group of drugs that includes antidepressants, anticonvulsants (antiseizure drugs), and others. These drugs have been shown to relieve pain independent of their effects on depression; that is, patients who are not depressed may experience pain relief.
Alternative causes of thrombocy to blood pressure normal child generic 50mg atenolol otc penia in critically ill patients include extracorporeal devices blood pressure classification chart purchase atenolol visa, intra-aortic balloon pumps arteria femoralis profunda cheap atenolol 50mg with visa, sepsis blood pressure ranges in pregnancy purchase atenolol with a visa, disseminated intravascular coagulation, bleeding, and medications. Suspected when a patient has a decrease in absolute platelet count to less than 150,000/mm3 or a relative decrease of at least 50%, skin lesions at injection sites, or systemic reactions after intravenous boluses. The typical onset is 5�10 days after heparin exposure, though it can be delayed and occur up to 3 weeks after cessation of therapy. Patients with recent unfractionated heparin/low molecular weight heparin exposure and new thrombosis should have their platelet counts checked before starting anticoagulant therapy. A higher antibody concentration leads to more color production and a higher optical density reading. Detects a range of immunoglobulin (Ig) A and IgM antibodies that are not pathogenic. Patient serum is mixed with washed platelets from healthy volunteers and low and high concentrations of heparin. Immediately discontinue all sources of heparin and initiate an alternative non-heparin anticoagulant. Parenteral direct thrombin inhibi to rs are associated with a higher rate of major bleeding complications compared with unfractionated heparin, and no antidote is available for excessive anticoagulation. Initiate warfarin once the platelet count has recovered and is within normal limits (at least 150,000/mm3) and after at least 5 days of therapy with an alternative anticoagulant. Alternatively, conservative warfarin dosing may begin once platelet count is recovering. Argatroban dosing in the critically ill population (Crit Care 2010;14:R90; Ann Pharmacother 2007;41:749-54) i. Bivalirudin dosing in the critically ill population (Pharmacotherapy 2006;26:452-60) i. Slightly higher doses may be necessary with continuous renal replacement therapy (0. A 93-year-old bedbound man (weight 45 kg) is admitted from a nursing home with a chronic obstructive pulmonary disease exacerbation requiring mechanical ventilation. Discontinue heparin drip, and initiate fondaparinux at 10 mg subcutaneously daily. The World Health Organization describes palliative care as �an approach that improves the quality of life of patients and their families facing the problems associated with life-threatening illness, through the prevention and relief of suffering by means of early identifcation and impeccable assessment and treatment of pain and other problems, physical, psychosocial, and spiritual� (Global Atlas of Palliative Care at the End of Life). Providers and families can collaborate to identify the sources of pain and relieve them with drugs and other forms of therapy. Symp to m management involves treating symp to ms other than pain such as nausea, thirst, bowel and bladder problems, depression, anxiety, dyspnea, and secretions. Emotional and spiritual support is important for both the patient and the family in dealing with the emotional demands of critical illness. Minimizing or discontinuing routine vital sign checks, patient weights, cardiac or other electronic moni to ring, fngersticks, and intermittent pneumatic compression devices. Consider discontinuing routine blood draws, radiologic imaging, and other diagnostic procedures. Opioids are the mainstay of treatment for patients experiencing pain at the end of life. Administer opioid as an intravenous bolus dose and begin an intravenous continuous infusion, adjusting rates to maintain comfort; avoid using subcutaneous or enteral routes because the onset is delayed. No evidence that unconscious patients do not experience pain; therefore, opioid administration should be initiated or continued d. Bolus and titrate infusions to control labored respirations; specifc dosages of medications are less important than the goal of symp to m relief. Optimal dose is determined by assessing the patient and rapidly increasing it as needed until symp to ms are no longer present. Dose determined by symp to m relief and adverse effects (excessive sedation, respira to ry depression [rare]). Suggested goals include keeping the respira to ry rate at or below 30 breaths/minute and eliminating grimacing and agitation. Symp to ms at the end of life can relate to acute or chronic anxiety, delirium, or terminal delirium. Nonpharmacologic treatments for agitation and anxiety can include frequent reorientation to the environment and reduction in noise and other bothersome or stimulating environmental fac to rs. Dose is determined by assessing the patient and increasing as needed until symp to ms are no longer present if haloperidol fails to relieve signifcant agitation. Determining what would be perceived as an acceptable level of sedation with the patient and/ or family or surrogate decision-maker is important before initiating sedatives. Acetaminophen is an effective therapy for improving comfort and decreasing the incidence of fever. A nonsteroidal anti-infamma to ry drug may be used when acetaminophen is ineffective. Dexamethasone, which is also known to have antipyretic properties, could be considered. Underlying causes such as medications, uremia, ascites, gastroparesis, and intestinal or gastric obstruction should be treated or eliminated, if possible. Agents to consider include haloperidol, me to clopramide, ondansetron, and dexamethasone. Excessive coughing can lead to exacerbation of dyspnea, and spells of nausea and vomiting, in addition to disturbing sleep and exacerbating pain. Non-opioid antitussives such as benzonatate and dextromethorphan may be considered. All opioids have intrinsic antitussive action by inhibiting the brain stem cough center; however, if the patient is on an opioid for other reasons, adding another opioid has not shown additional beneft. Near the end of life, the ability to clear oral and tracheobronchial secretions diminishes. Secretions are usually to o low in the tracheobronchial tree for gentle oral suctioning to be of help, and suctioning can be disturbing. The mainstay of treatment includes anticholinergic and antimuscarinic medications. Scopolamine and atropine cross the blood-brain barrier and can be more sedating than glycopyrrolate. Speaking with the patient, you fnd that she wishes not to be resuscitated or intubated but only to be comfortable. In a meeting with the patient�s family, all members agree that they do not want to see their loved one suffer any longer. Titration parameters include giving a bolus dose equivalent to the current rate and increasing the infusion by 25%. The nurse taking care of the patent believes that the titration parameters are to o aggressive. Change the parameters to increase only the morphine drip when the patient shows signs of discomfort, such as an increase in blood pressure or heart rate. Discontinue titration parameters, keeping the morphine infusion at the current rate. Discontinue titration parameters, keeping the morphine infusion at the current rate and adding a midazolam infusion at 2 mg/hour. Give your patient a fast hug (at least) prophylaxis in the critically ill: a meta-analysis. Surviving Stress Ulcer Prophylaxis Sepsis Campaign: international guidelines for 1. Pro to n management of severe sepsis and septic shock: pump inhibi to rs versus histamine 2 recep to r 2012. Effect of histamine-2 prophylaxis with pro to n pump inhibi to rs, H2 recep to r antagonists versus sucralfate on stress ulcer recep to r antagonists. Am J of pro to n pump inhibi to rs vs histamine-2 recep to r Gastroenterol 2012;107:507-20. Crit of stress ulcer prophylaxis and risk of nosocomial Care Med 2010;38:1197-1205. A comparison inhibi to r for stress ulcer prophylaxis in critically ill of sucralfate and ranitidine for the prevention of patients. Canadian Critical Care recep to r antagonists vs pro to n pump inhibi to rs on Trials Group. Crit Care Med H2-antagonists for stress ulcer prophylaxis: a meta 1999;27:2812-7.


